WHY THIS MATTERS IN BRIEF
Being able to watch cells move under the microscope isn’t the same as being able to observe them in their natural environment, and this breakthrough will lead to a better understanding of cell biology and help create new revolutionary medical treatments.
In 2016 we observed atoms interacting with each other for the first time, and next time you’re having a bad day, feeling sluggish, or simply uninspired about your general existence, today I have something that should help you get your mojo back. Watch the video below, and remember that within us all are fiery, dancing cells working their tiny little butts off just to keep us all alive.
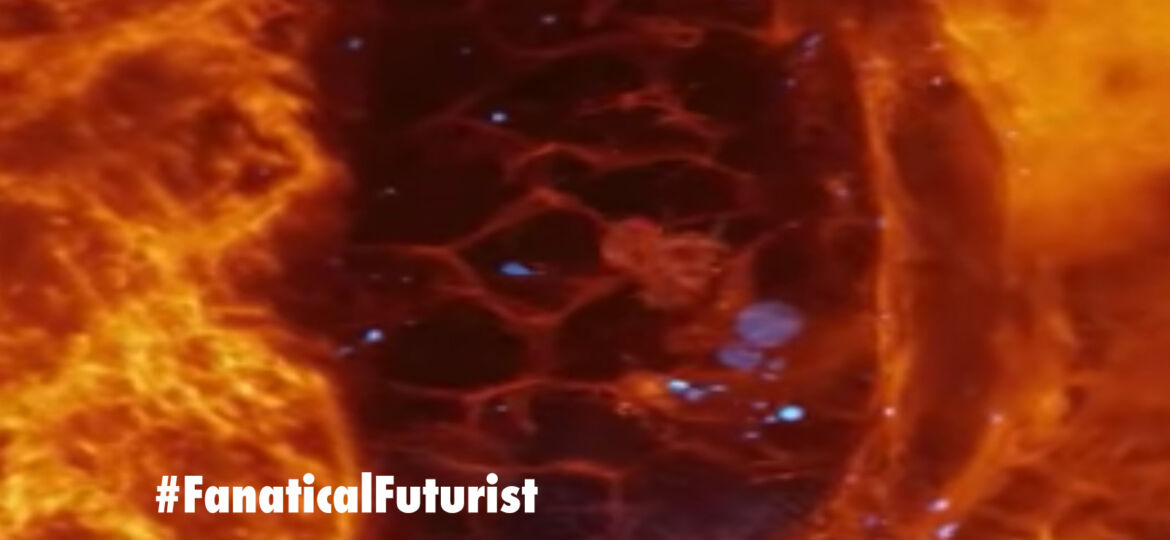
Although we’ve known for decades how cells move throughout organisms, this video is the first high resolution, 3D footage of that process as it’s happening, and as you can see it’s both freaky and amazing at the same time. For example, this video shows in real time an immune cell migrating through the inner ear of a tiny zebrafish, will wonders ever cease…
The blue dots you can see the immune cell swallow up are particles of Dextran, a sugary polysaccharide that’s found in everything from wine and heart medication to dental plaque. In addition to being absolutely mesmerising, this footage is exciting because it’s the first of its kind, and it was done using a brand new type of microscopy.
Sure, we’ve seen cells under the microscope for hundreds of years, but when we try to get footage of them moving, the results are at best fuzzy and at worst utter rubbish, so our clearest images have always come from groups of cells preserved on glass slides which meant we were never ever getting the full story. And now with this “fly on the wall” documentary style technology we can.
“This raises the nagging doubt that we are not seeing cells in their native state, happily ensconced in the organism in which they evolved,” says team leader and physicist Eric Betzig, from the Howard Hughes Medical Institute in Virginia who performed the work, “even when we try to see just one cell at a time the cells are blasted with intense light and our microscopes are too slow to follow all the action in 3D. This also contributes to our fear that we are not seeing cells in their natural, unstressed form. It’s often said that seeing is believing, but when it comes to cell biology, I think the more appropriate question is, ‘When can we believe what we see?'”
To overcome this, Betzig and his team combined two microscopy technologies, namely adaptive optics, and lattice light sheet microscopy.
Adaptive optics is the technology astronomers use to see distant celestial objects through Earth’s rippling atmosphere, and in the case of imaging a living organism, this means shining a laser on whatever the researchers want to image, and using this to measure how much light distorts as it passes through surrounding cells and tissues.
They then counteract for these interruptions with equal but opposite light distortions, to allow greater visibility in their target area in a similar to how noise-cancelling headphones work.
The result? A totally clear image of a cell in its natural environment.
The second technique, lattice light sheet microscopy, lets the researchers capture those super-clear images in real time, and it involves rapid and repeated sweeps of ultra-thin sheets of light, which creates a series of 2D images that can be built into a moving high-res 3D picture, all without frying the cell or interfering with its activity.
The result is something scientists have wanted for years – bright, clear, and vibrant images of our cells in action under the microscope, and there’s no doubt that this breakthrough is going to lead to discoveries we can’t even imagine right now.
For example, here’s a bad old cancer cell trailing sticky appendages as it rolls through a blood vessel and attempts to hook in somewhere on the vessel wall… and now, I’ll wager, thanks to this technology you’re already seeing cancer in a new light, and that’s just the beginning.
The main caveat for all this though is that right now this microscopy set up is incredibly expensive and not particularly user friendly. For example, the microscope Betzig and his team uses takes up a 3 metre (10 ft) long table. But now that we have the proof of concept, the team predicts it’s only a matter of time before this technology revolutionises our understanding of biology and human cells.
The team is already working on a smaller and cheaper next generation version of the microscope, and they’ve also said they’ll share the plans for free so other labs can make their own versions.
“It’s a bit of a Frankenstein’s monster right now,” says Betzig, “if you really want to understand the cell in vivo, and image it with the quality possible in vitro, this is the price of admission.”
For now though just bask in the awesomeness of the little guys, the cells in your body, that are keeping you alive right now.