WHY THIS MATTERS IN BRIEF
- HIV is a stubborn virus and while scientists have managed to “rip” it out of patients noone has yet managed to create a vaccine for it, we’re hoping that is about to change
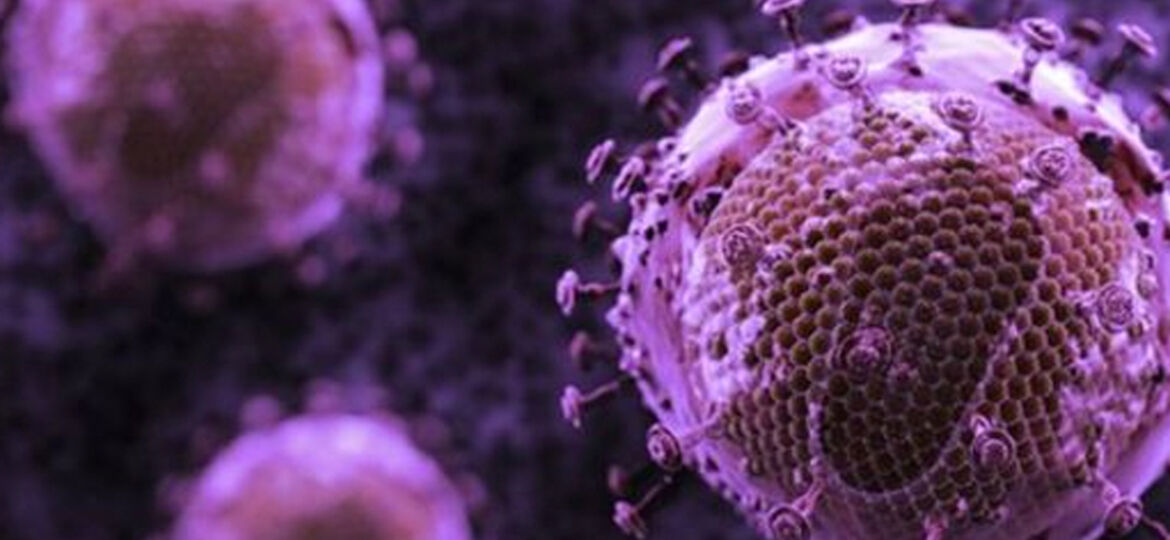
HIV is a stubborn disease but over the past year scientists have been making progress against it, even going so far as to rip it right out of a living patients cells and now researchers in Canada believe they may have gone one step better and may be on the cusp of creating the worlds first HIV vaccine. It’s passed the first of three standard testing stages and it’s on the cusp of being ready for human trials across North America next year.
Dr. Chil-Yong Kang and a team of researchers developed the vaccine, named SAV001, at Western’s Schulich School of Medicine and Dentistry.
Dr. Kang was first invited to work on the vaccine in 2006 by the Canadian branch of the Korean pharmaceutical company Sumagen and the first phase of SAV001’s testing involved administering it to 33 HIV-positive patients to make sure it didn’t have any adverse side effects.
“We demonstrated that it is completely safe, and that it induces the proper immune-responses. So in a way, the phase one result predicts the outcome of the phase two trial,” Kang said.
The drug works by producing antibodies in the patient that neutralize many different strains of the virus, which are the immune responses the drug displayed in the first phase.
The second phase will include 600 subjects from across North America, half of whom are general volunteers, and half of whom are high-risk but HIV-negative. It will check to see if the drug produces the desired effect to a significant extent.
High-risk individuals include intravenous drug users and people living with HIV-positive partners.
If the drug passes through the second phase, it will be administered to 6,000 people across the globe as part of the final stage of testing. At this stage, vaccines will be tested to determine if the drug can significantly reduce the rate of contraction, and if so then the drug will move on to be mass produced.
SAV001 is the worlds first preventative “killed-whole” vaccine, meaning that it contains a genetically engineered version of the virus that is safe and can be produced in large quantities – this is the method used for Polio, Rabies, Hepatitis-A and the Flu vaccines, but it has never been done successfully in an HIV vaccine.
It is also the first successful killed-whole HIV vaccine to be approved by the US Food and Drug Administration (FDA) for human testing and that’s why the drug also happens to be highly controversial. SAV001 is a polarising development in what Kang dubbed a “highly competitive” area of research.
“Some people said ‘this is innovative, go ahead and publish.’ But some people were highly critical,” said Kang. “There are maybe 100 HIV vaccine trials going on right now, but so far I have not seen any promising results. And no one has tried our strategy.”
Killed-whole vaccines can be dangerous, as the form of the virus within the drug may not be entirely killed. Kang and his team solved both of these problems by neutralising the virus with both chemicals and radiation and Western has a patent protecting the new method as its IP.
There have been other prominent HIV vaccines to pass through to the final testing stage, though none have been of this type and if everything runs smoothly, the second stage of the testing will begin in fall of 2017.
While SAV001 is a preventative vaccine, and meant to be taken to prevent the virus from being contracted, Kang is also working on a therapeutic vaccine meant to be taken after an individual has already been infected that works by educating the patient’s white blood cells to help them to destroy the cells that HIV infects, such as T-Cells. And again this particular drug differs from other therapeutic HIV medications because while most drugs mitigate the progression of the virus, the virus begins progressing again if the medication is stopped.
If Kang’s vaccine is effective, it will destroy the patient’s virus entirely, providing a solution to the 37 million people globally already infected with HIV and AIDS.
The vaccine has already completed testing in small animals and will begin testing on medium sized animals in January 2017.