
WHY THIS MATTERS IN BRIEF
Life’s DNA has always been coded using just four letters, but now researchers have added two new letters and it opens the door to a whole new world of alien lifeforms never seen before.
Ever since life gained a foothold on this fragile rock three and a half billion years ago its story has been written in a DNA code consisting of just four letters – G, T, C and A, that all pair up to form the DNA double helix, and the sequence of these letters, up until now, has determined the makeup and characteristics of every organism on Earth – from the earliest sponges and Amonites to the dinosaurs… and us.
Over the decades adding, deleting, and splicing genes has become routine, and some researchers are now even designing new DNA to create new creatures, such as E. coli that are resistant to every known virus on Earth. But while many researchers are hard at work rearranging the existing DNA letters into new sequences a new experiment run by the Scripps Research Institute in California has redefined the concept of synthetic biology by creating and writing two new letters.
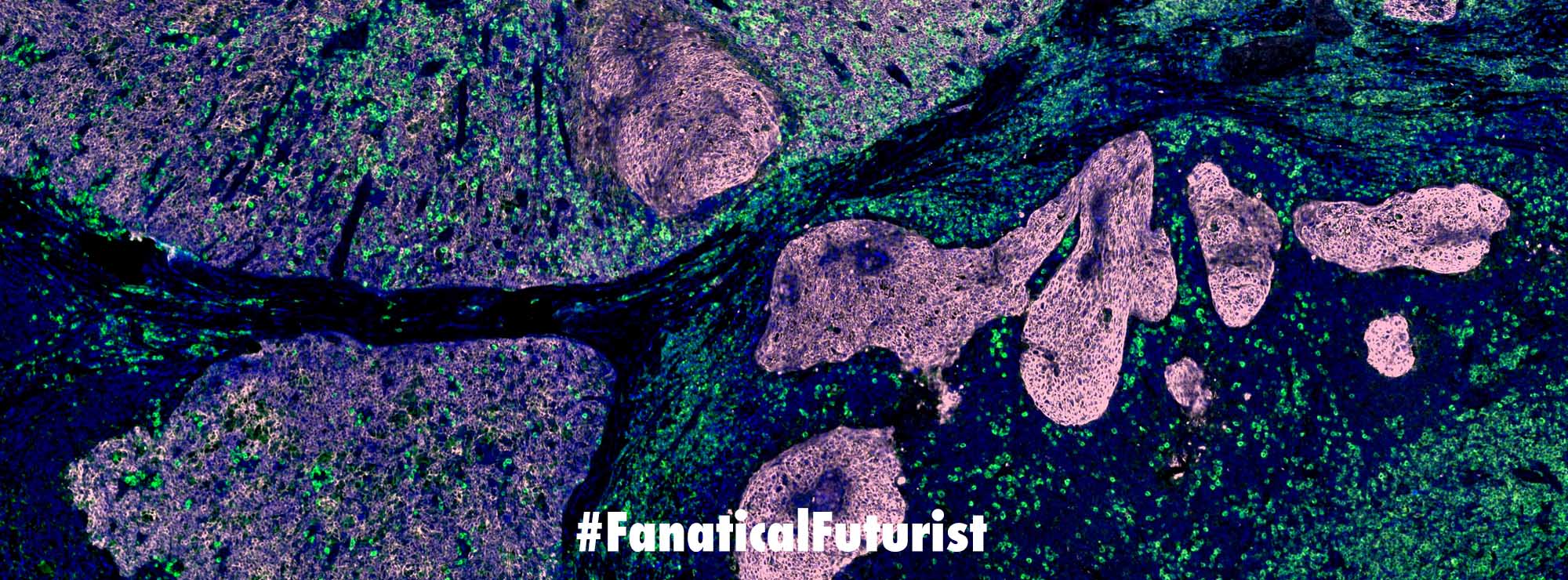
On Monday the team at Scripps, led by Floyd Romesberg announced in a paper published in Nature that they have expanded the genetic alphabet of DNA from four to six with the addition of two new letters, X and Y. And they showed that the new letters could be integrated into the DNA of a living creature, in this case an E. coli bacterium – exponentially increasing the amount of information the genetic code can store, bearing in mind that just a shoebox of existing DNA could be used to store all the world’s information.
“This is a very major accomplishment in our efforts to inch towards creating a completely synthetic biology,” says Steven Benner, a synthetic biologist at the Foundation for Applied Molecular Evolution who was not involved in the study, “many in the broader community thought that Floyd’s result would be impossible to achieve.”
The history of the two new letters can be traced back to 1998 when Rosmeberg and his colleagues first tinkered with the idea. They wanted to create a pair of genetic letters that were similar enough to the letters life already uses so that they could function, and wouldn’t be rejected by, the existing DNA framework. But the letters had to be different enough that they wouldn’t be accidentally mispaired with the original four letters and effectively forgotten.
The letters saw many, many failed incarnations before they morphed into what the researchers presented today.
“After 14 years we had developed, made, and optimized over 300 of these nucleotides,” Rosmeberg says. Then came the bigger hurdle – getting a real cell to reproduce X and Y after the scientists had spliced them in. While cells come stocked with the machinery to churn out the original four letters, the scientists had no way to easily alter their bacteria to likewise synthesize X and Y.
Over time Romesberg’s team found a way to steadily feed their bacterium the X and Y nucleotides it would need. They took a line of genetic code from a species of fungus – which saves energy by gobbling up nucleotides anywhere around it and using those when it needs to replicate its own DNA – and spliced it into their bacteria’s loop of DNA.
“As soon as we cut in that DNA, it was remarkable. The issue of producing these nucleotides just vanished as a problem,” Romesberg says.
As long as their bacteria was fed the X and Y nucleotides, the researchers found that even after 15 hours it would replicate them over and over again just as it would any other line of DNA. And if the bacteria tried to reproduce without them, it would get stuck until it mutated out the new code, essentially reverting back to its natural state.
It’s an open question why the bacteria cell doesn’t reject the strange nucleotides as fatal errors in genetic replication – the genetic framework of life has many inspection and failsafe mechanisms to prevent errors or foreign debris from gunking up DNA. Romesberg hypothesizes that his expanded genetic alphabet is either so foreign that the genetic framework simply doesn’t recognize it as an error, or, more likely, it does see an error but has no way to fix or change the X and Y letters.
Understandably, there’s growing concern about these kinds of ultra modified organisms, these new forms of life, and what would happen if they were to somehow escape from the lab.
“I’m worried people might think we’re making Frankenstein,” Romesberg says, “but I’d like to emphasize that we have a failsafe mechanism built into this system. If you take this bacteria out of the lab it converts back to its normal state.”
Although Rosemberg’s modified E. coli replicated his X and Y nucleotides in the DNA, the bacteria did not use that customized clip of genetic code to build proteins, so the new letters were not expressed in new genes. While DNA stores genetic information in its double helix, it is not until that info is transcribed to RNA that the cell begins to use it in three-letter chunks called codons. This was intentional, Rosemberg says. His X and Y nucleotides are hidden away on a length of DNA that essentially functions as empty code.
But the prospect of using these new nucleotides to craft new codons is perhaps the most exciting aspect of this discovery. While all possible combinations of G, T, C, and A are already in use – AAG for example, creates an amino acid called lysine, and TAA signifies the end of a code of DNA – new letters exponentially increase the number of possible codons and give researchers the ability to recode the genetic framework without needing to rewrite or erase what life has already created. Codons like XYA or TGX, for example, could be programmed to build new types of amino acids, which could configure new proteins, and they could even be coded to create new materials.
Expanding the suite of possible cell-made proteins would have an immediate impact in the pharmaceutical industry, which is increasingly using the protein synthesis of cells – rather than step by step molecular configuration – to produce medicines. And while this sounds like little more than science fiction, researchers have already proven the ability to recode how DNA is expressed with the original four nucleotides.
“There is still much to do to achieve such goals, but this is an exciting early step,” says Sri Kosuri, a molecular biologist at UCLA.
We are at the very beginning of creating whole new forms of life, unlike anything this rock has seen before. In the movie Prometheus the alien humanoids were special, and “transcendent” because their DNA was superior to our own, and it was much more dense and capable of carrying much more information and genetic richness. This new breakthrough has echoes of that storyline. Darwin is dead, the genie is out the box – who knows where this dance ends…














How does the failsafe work? How does it detect when it’s removed from the lab, and react to it?