WHY THIS MATTERS IN BRIEF
Now that we have access to advanced gene editing techniques, such as CRISPR, it is inevitable that we will progressively use these technologies to edit the human genome and create genetically altered human beings.
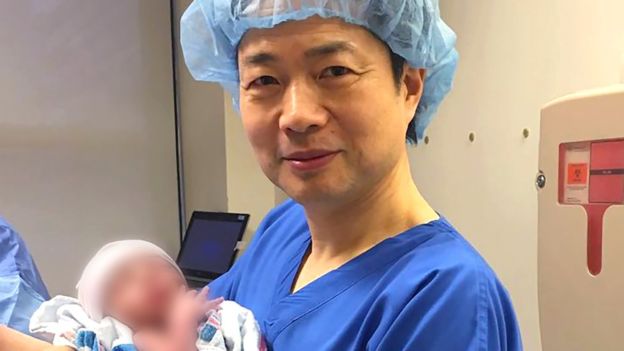
The world’s first baby to be born from a new procedure that combines the DNA of three people appears to be healthy, according to doctors in the US who oversaw the treatment.
The baby was born on 6 April after his Jordanian parents travelled to Mexico where they were cared for by US fertility specialists.
Baby “x”
Doctors led by John Zhang, from the New Hope Fertility Center in New York, decided to attempt the controversial procedure of mitochondrial transfer in the hope that it would give the couple a healthy child. While many experts welcomed news of the birth, some raised concerns that the doctors had left the US to perform the procedure beyond the reach of any regulatory framework and without publishing details of the treatment.
Speaking about the event Zhang said he went to Mexico where “there are no rules” and insisted that doing so was right.
“To save lives is the ethical thing to do,” he said.
Mitochondrial transfer was legalised in the UK in 2015 but so far no other country has introduced laws to permit the technique. The treatment is aimed at parents who have a high risk of passing on debilitating and even fatal genetic diseases to their children.
The boy’s mother carries genes for the fatal Leigh Syndrome, which harms the developing nervous system. The faults affect the DNA in mitochondria, the tiny battery-like structures that provide cells with energy, and are passed down from mother to child.
Ten years after the couple married, the wife became pregnant but she lost the baby in the first of four miscarriages. The couple had a baby girl in 2005 who died at the age of six, and later, a second child who lived for only eight months. Tests on the wife showed that while she was healthy, about one-quarter of her mitochondria carried the genes for Leigh syndrome.
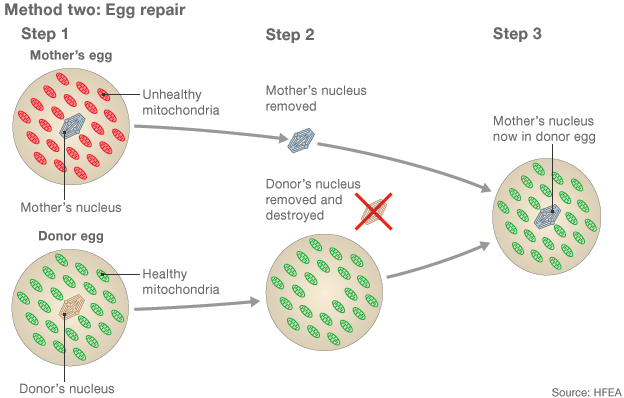
When the couple approached Zhang for help, he decided to try the mitochondrial transfer procedure. He took the nucleus from one of the woman’s eggs and inserted it into a healthy donor’s egg that had had its own nucleus removed. He then fertilised the egg with the husband’s sperm.
The US team created five embryos but only one developed normally. This was implanted into the mother and the baby was born nine months later.
The baby is not the first child to be born with DNA from three people. In the 1990s, fertility doctors tried to boost the quality of women’s eggs by injecting cytoplasm, the cellular material that contains mitochondria, from healthy donor eggs. The procedure led to several babies being born with DNA from the parents plus the healthy donor. Some of the children developed genetic disorders and the procedure was banned.
Speaking about the latest case, Dusko Ilic, a stem cell scientist at King’s College London, said: “Without much ado, it appears the first mitochondrial donation baby was born three months ago. This was an ice-breaker. The baby is reportedly healthy. Hopefully, this will tame the more zealous critics, accelerate the field, and we will witness soon the birth of the first mitochondrial donation baby in the UK.”
But some questions remained, he said.
“By performing the treatment in Mexico, the team were not subject to the same stringent regulation as some other countries would insist on. We have no way of knowing how skilful or prepared they were, and this may have been a risky thing to do.
“On the other hand, we have what appears to be a healthy baby. Because it was successful, fewer questions will be raised, but it is important that we still ask them.
“Was this the first time ever they performed the technique or were there other attempts and they are reporting this one because it was successful?
“This and other important questions remain unanswered because this work has not been published and the rest of the scientific community has been unable to examine it in detail. It’s vital that that happens soon.”
Alison Murdoch, a fertility doctor at Newcastle University, said: “If this baby has been born as suggested then that would be great news. The translation of mitochondrial donation to a clinical procedure is not a race but a goal to be achieved with caution to ensure both safety and reproducibility.”
Details of the birth are due to be presented at the American Society for Reproductive Medicine meeting in Salt Lake City in October.
Doug Turnbull, a neurology professor at Newcastle University who pioneered mitochondrial transfer in the UK, said the technique offered hope to mothers who carried mitochondrial DNA mutations.
“There have been extensive discussions in the UK to ensure that families with mitochondrial disease get the best possible advice about their reproductive options and that any new IVF-based technique is appropriately regulated and funded. This abstract gives very little information about the technique used, the follow up of the child or the ethical approval process.”