WHY THIS MATTERS IN BRIEF
3D printing could give infertile couples new hope.
Northwestern University scientists have created a prosthetic ovary using a 3D printer – an implant that allowed mice that had their ovaries surgically removed to bear live young and the results will be presented at the Endocrine Society’s annual meeting, ENDO 2016, in Boston.
Researchers are hopeful that they can use the technology to develop an ovary bioprosthesis that can eventually be implanted in women to restore fertility and one group that could benefit is survivors of childhood cancers, who have an increased risk of infertility as adults. An estimated 1 in 250 adults has survived childhood cancer.
“One of the biggest concerns for patients diagnosed with cancer is how the treatment may affect their fertility and hormone health,” said lead study author Monica M. Laronda, PhD, a postdoctoral research fellow at Northwestern University’s Feinberg School of Medicine.
“We are developing new ways to restore their quality of life by engineering ovary bioprosthesis implants.”
How they did it
The researchers used a 3D printer to create a scaffold to support hormone-producing cells and immature egg cells, called oocytes. The structure was made out of gelatin – a biological material derived from the animal protein collagen. The scientists applied biological principles to manufacture the scaffold, which needed to be rigid enough to be handled during surgery and to provide enough space for oocyte growth, blood vessel formation and ovulation. Using human cell cultures, the researchers determined the optimal scaffold design should have crisscrossing struts that allowed the cells to anchor at multiple points. The scaffolds were seeded with ovarian follicles – the spherical unit that contains a centralized oocyte with surrounding supportive, hormone-producing cells – to create the bioprosthesis.
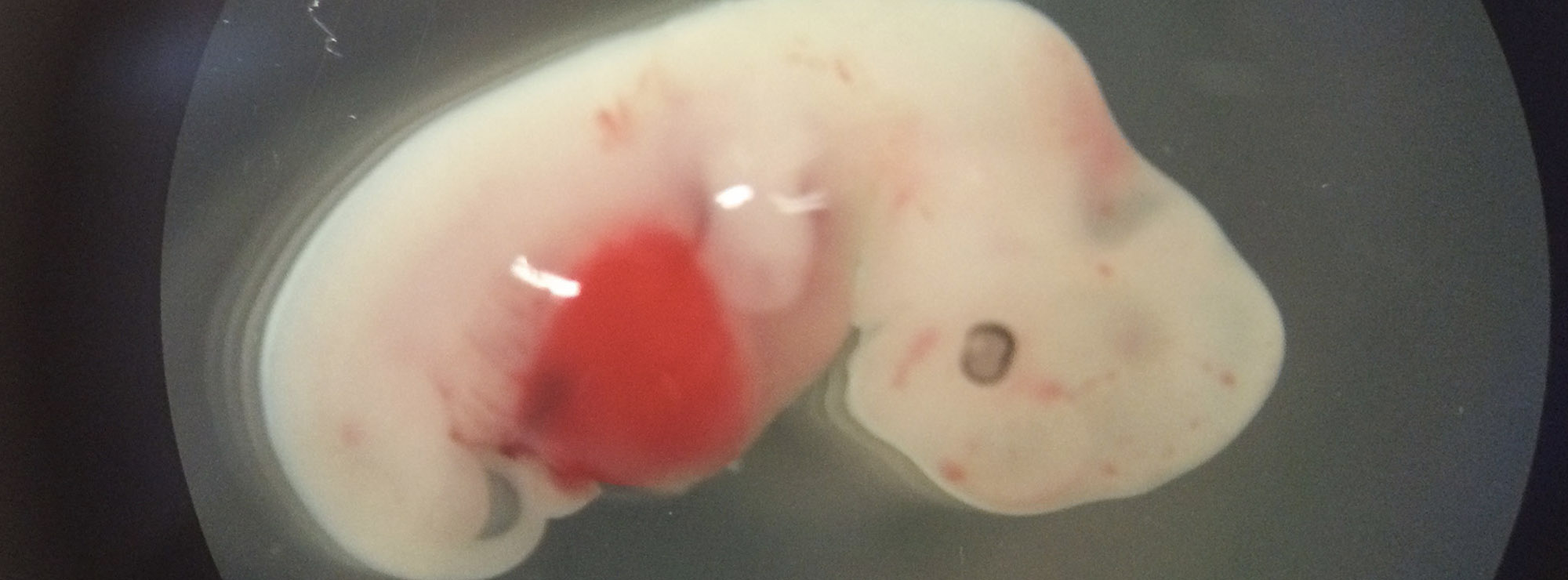
To test the implant, researchers removed the ovaries of mice and replaced them with the ovary bioprosthesis. Following the procedure, the mice ovulated, gave birth to healthy pups and were able to nurse.
Implanting the prosthetic ovary in mice also restored the estrous, or female hormone cycle. Researchers theorize a similar implant could help maintain hormone cycling in women who were born with or have undergone disease treatments that have reduced ovarian function. These women often experience decreased production of reproductive hormones that can cause issues with the onset of puberty as well as bone and vascular health problems later in life.
“We developed this implant with downstream human applications in mind, as it is made through a scalable 3D printing method, using a material already used in humans,” Laronda said.
“We hope to one day restore fertility and hormone function in women who suffer from the side effects of cancer treatments or who were born with reduced ovarian function.”
The implant’s scaffold structure supported the growth of blood vessels in the mice without the use of any substances to stimulate the process. Using manufacturing techniques inspired by human biology, the researchers developed a scaffold that interacts with the body’s tissues. This process could influence future work on complex soft tissue replacement, Laronda said.
The National Institutes of Health, the Burroughs Wellcome Fund Career Award at the Scientific Interface, and the National Science Foundation Graduate Research Fellowship Program provided support for the research.